| Networked Closed-loop Medical Cyber-Physical Systems (MCPS) |
BackgroundCritical care involves the use of two common types of medical devices: monitoring devices, such as bedside monitors; and actuation devices, such as infusion pumps. Traditionally, caregivers need to collect information from the monitoring devices and manually adjust the settings of actuating devices. Patient safety may be compromised if a caregiver is diverted by an emergency and misses some important events. Physiological closed-loop interoperable systems have been proposed to solve this problem. In such a system, a controller is networked with sensors and actuators to continuously monitor the patient's physiological states and automatically control the actuators within the safe operation region, thereby partially closing the loop. Caregivers will be alerted if the patient's states become unstable. 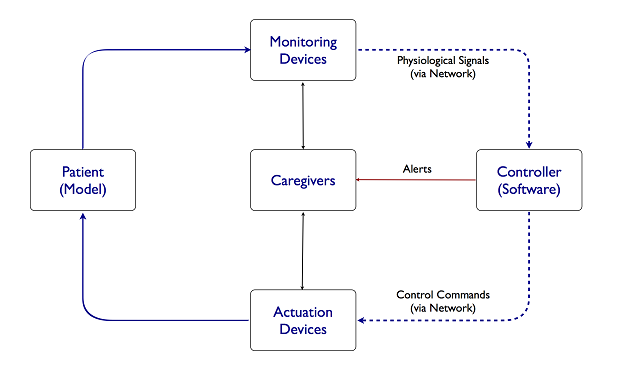
Safety AnalysisPatient safety is the primary concern in closed-loop MCPS. The complex interaction between the continuous time physiological dynamics of patients and the discrete, uncertain nature of networked control systems poses a major challenge on design and verification. We have developed a model-driven approach to prove the safety properties of MCPS. Our method combines simulation-based analysis of a detailed model of the system (including continuous-time patient dynamics) with model checking of an abstract timed-automata model. We have shown that crucial timing behavior is preserved across the two models, which ensures the conservativeness of the safety analysis. We have also proposed a system design that can guarantee open-loop safety under network failure. We have applied this approach to the development of a closed-loop Patient-Controlled Analgesia (PCA) system to prevent overmedication. We are currently working on extending the framework to more complicated clinical use cases such as closed-loop blood glucose regulation. In-silico Evaluation and Enhancement of Hospital Insulin Infusion ProtocolsIn-patient glycemic control, particularly in critically ill patients, remains challenging. Patients with impaired insulin sensitivity and/or secretion (i.e. type 1 diabetes mellitus) often manifest extremely labile blood glucose measurements during such periods of stress and inflammation. Most current insulin infusion protocols are developed based on clinical experiences and consensus among a local group of physicians. Recent advances in human glucose metabolism modeling have established a computer model that invokes algorithms representing many of the pathways involved in glucose dysregulation for patients with diabetes. The model-based closed-loop MCPS framework can be applied to build safe and effective glucose control systems. In this collaborative case study with critical care clinicians, we used an FDA approved glucose metabolism model to evaluate an existing institutional intraoperative insulin infusion protocol via closed-loop simulation on the virtual diabetic population that comes with the computer model. A comparison of simulated responses to actual retrospective clinical data from 57 type 1 diabetic patients undergoing cardiac surgery managed by the institutional protocol was performed. We then designed a proportional-derivative (PD) controller that overcomes the weaknesses exhibited by our old protocol while preserving its strengths. We proposed a method to optimize the controller on the high-dimensional, non-linear physiological model with uncertain parameters and states, for which the classical analytical controller tuning methods cannot be applied. In-silico evaluation results show that the new proportional-derivative controller more effectively manages intraoperative hyperglycemia while simultaneously reducing hypoglycemia and glycemic variability. By performing in-silico simulation on intraoperative glucose and insulin responses, robust and seemingly efficacious algorithms can be generated that warrant prospective evaluation in human subjects. Assurance CasesAn assurance case is a formal structure for organizing arguments and evidence to support the validity of a top-level claim. It is a useful mechanism for presenting and reviewing information in the medical device regulatory process. In the traditional "pair-wise" regulation process, the entire system needs to be re-regulated with the addition or replacement of any individual device. Alternatively, in the "component-wise" regulation paradigm, only the newly added or replaced device needs to be assessed as being compliant to the expected interface. We are developing a modular assurance case framework for closed-loop infusion systems. A modularized system assurance case integrates the assurance case modules for each system component and it facilitates the "component-wise" regulation. Current Members
Publications
|
